 
Paracetamol ( acetaminophen) is involved in around 40% of intentional overdose episodes, and remains the leading cause of acute liver failure in many countries including the United Kingdom. Immediate management of drug overdose relies upon the patient account of what was ingested and how much. Hewett, David G Shields, Jennifer Waring, W Stephen Missed paracetamol ( acetaminophen) overdose due to confusion regarding drug names. In patients with acetaminophen toxicity who co-ingest other medications that may potentially delay gastric emptying or in those with risk factors for delayed absorption of acetaminophen, we recommend close monitoring of aminotransferase enzyme levels, as well as trending acetaminophen concentrations until undetectable before discontinuing the antidote therapy. Delay in acetaminophen absorption may be associated with a second peak in serum concentration following an initial declining trend, especially in cases of concomitant ingestion of opioids. Prolongation of N-acetylcysteine infusion beyond the standard protocol, despite a significant gap in treatment, was critical for successful avoidance of hepatotoxicity. 
Here, we describe the case of a 65-year-old male with acetaminophen overdose and opioid co-ingestion who developed a second peak in acetaminophen serum levels after completing the recommended 21-hour intravenous N-acetylcysteine protocol and when the standard criteria for monitoring drug levels was achieved. Despite evidence on the efficacy of N-acetylcysteine for prevention of hepatic injury, controversy persists about the optimal duration of the therapy. N-Acetylcysteine is the recommended antidote for acetaminophen poisoning. Acetaminophen overdose, a frequent cause of drug toxicity, has been recognized as the leading cause of fatal and non-fatal hepatic necrosis. Īcetaminophen overdose associated with double serum concentration peaksįull Text Available Acetaminophen is the most commonly used analgesic–antipyretic medication in the United States. a vein) A laxative Medicine to reverse the effects of the poison and treat symptoms Tube through. Acetaminophen and codeine overdose occurs when someone takes. and is not helped by other types of painkillers. APAP-CYS concentrations varied substantially among the patients with acetaminophen toxicity (0.10 to 27.3 nmol/ml. The mean (SD peak APAP-CYS concentrations for the Trials were: Trial 1- 0.4 (0.20 nmol/ml, Trial 2- 0.1 (0.09 nmol/ml and Trial 3- 0.3 (0.12 nmol/ml. In addition, we collected samples from 19 subjects with acute acetaminophen ingestion, 7 subjects with repeated acetaminophen exposure and 4 subjects who ingested another hepatotoxin. Results Trial 1 included 144 samples from 24 subjects Trial 2 included 182 samples from 91 subjects and Trial 3 included 200 samples from 40 subjects. Serum APAP-CYS was measured using high pressure liquid chromatography with electrochemical detection. Patients in the observational study were categorized by type of acetaminophen exposure (single or repeated. Trial 1 consisted of non-drinkers who received APAP for 10 days, Trial 2 consisted of moderate drinkers dosed for 10 days and Trial 3 included subjects who chronically abuse alcohol dosed for 5 days. Methods Samples were collected during three clinical trials in which subjects received 4 g/day of acetaminophen and during an observational study of acetaminophen overdose patients. The objective of this study is to describe APAP-CYS concentrations in these clinical settings as well as to further characterize the concentrations observed following acetaminophen overdose. However, the concentrations of APAP-CYS during therapeutic dosing, in cases of acetaminophen toxicity from repeated dosing and in cases of hepatic injury from non- acetaminophen hepatotoxins have not been well characterized. 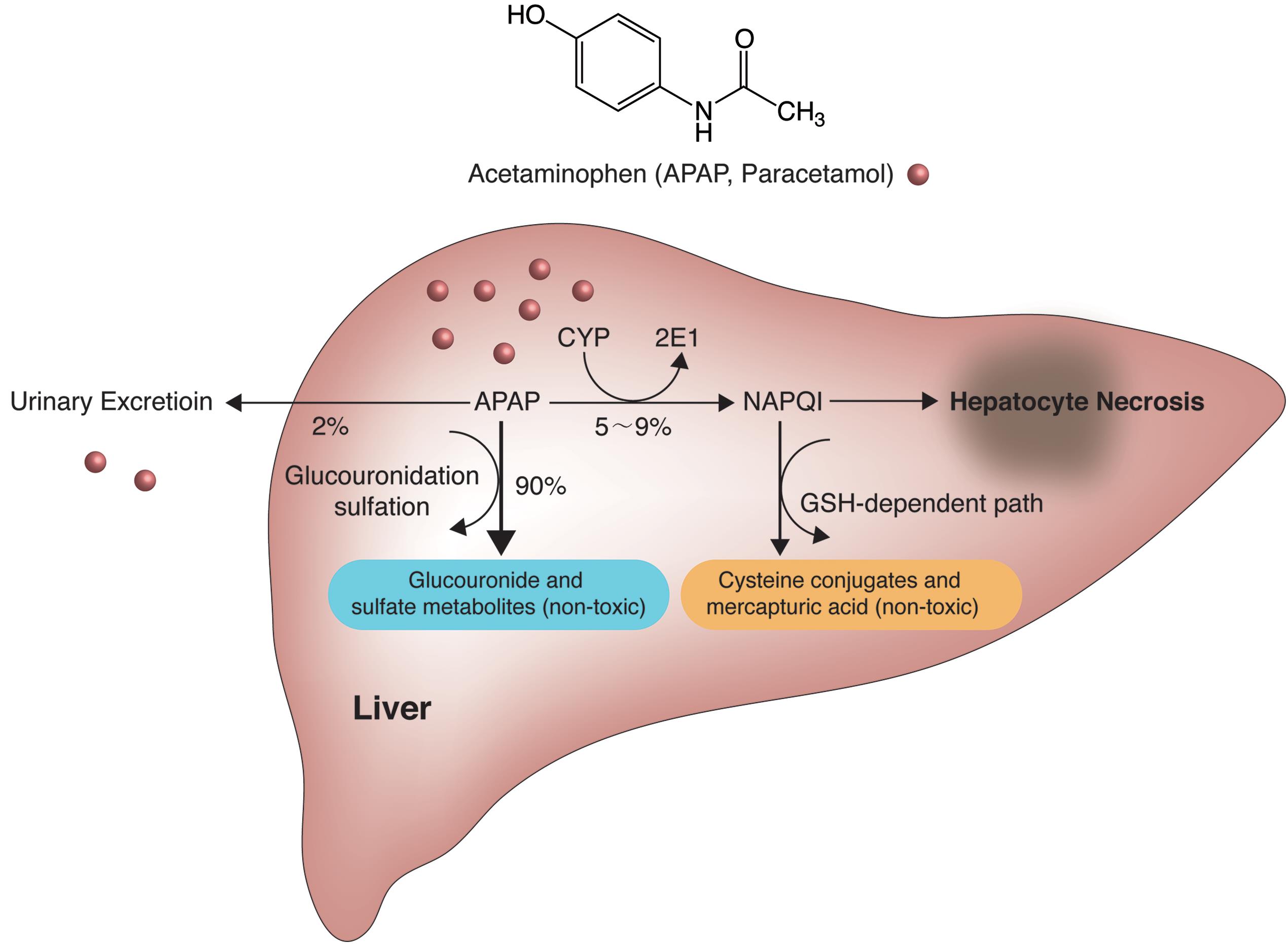
APAP-CYS concentrations have been described in the setting of acute overdose, and a concentration >1.1 nmol/ml has been suggested as a marker of hepatic injury from acetaminophen overdose in patients with an ALT >1000 IU/L. Acetaminophen-cysteine adducts during therapeutic dosing and following overdoseĭirectory of Open Access Journals (Sweden)įull Text Available Abstract Background Acetaminophen-cysteine adducts (APAP-CYS are a specific biomarker of acetaminophen exposure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
